Better data
for better decisions
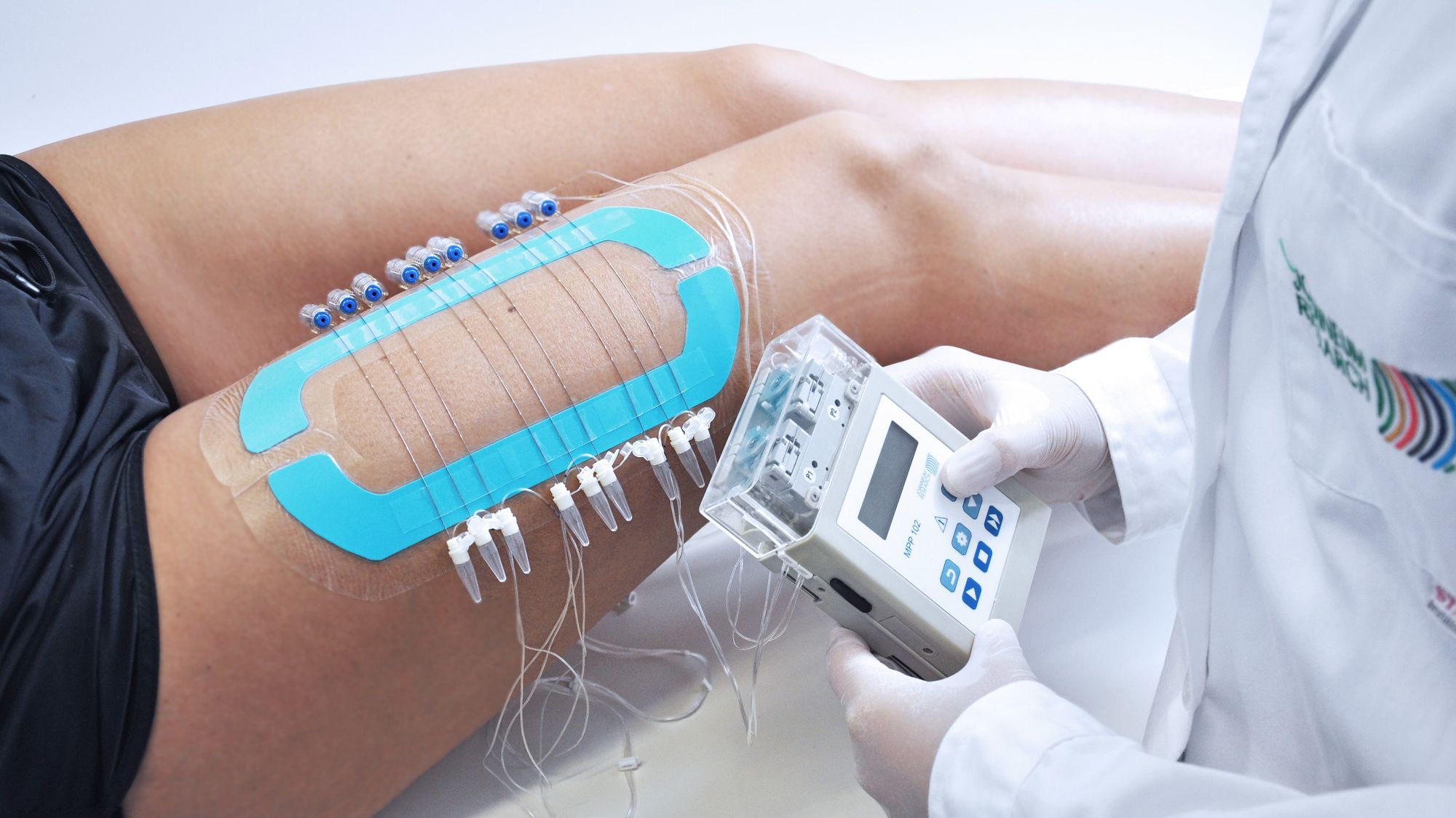
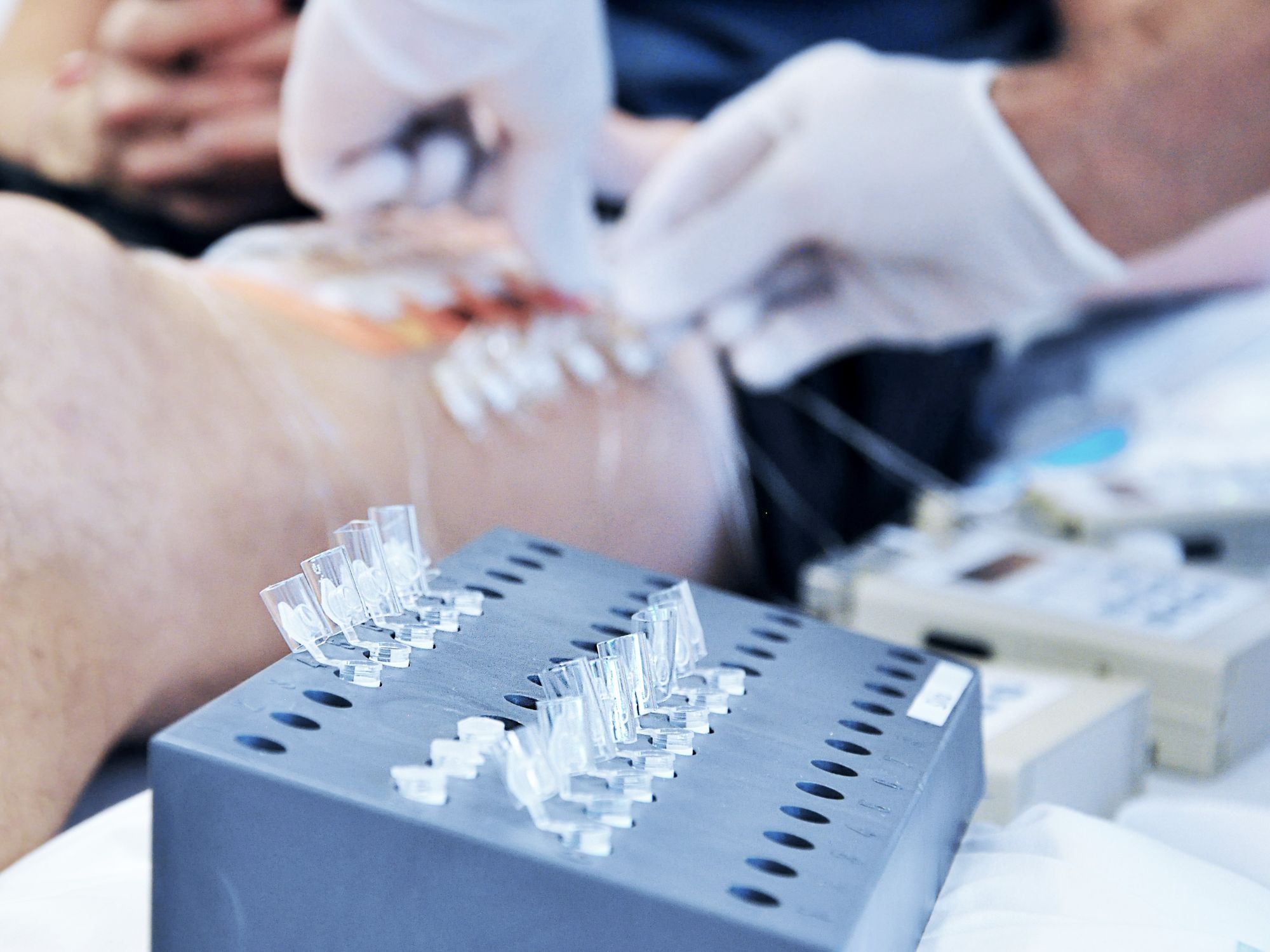
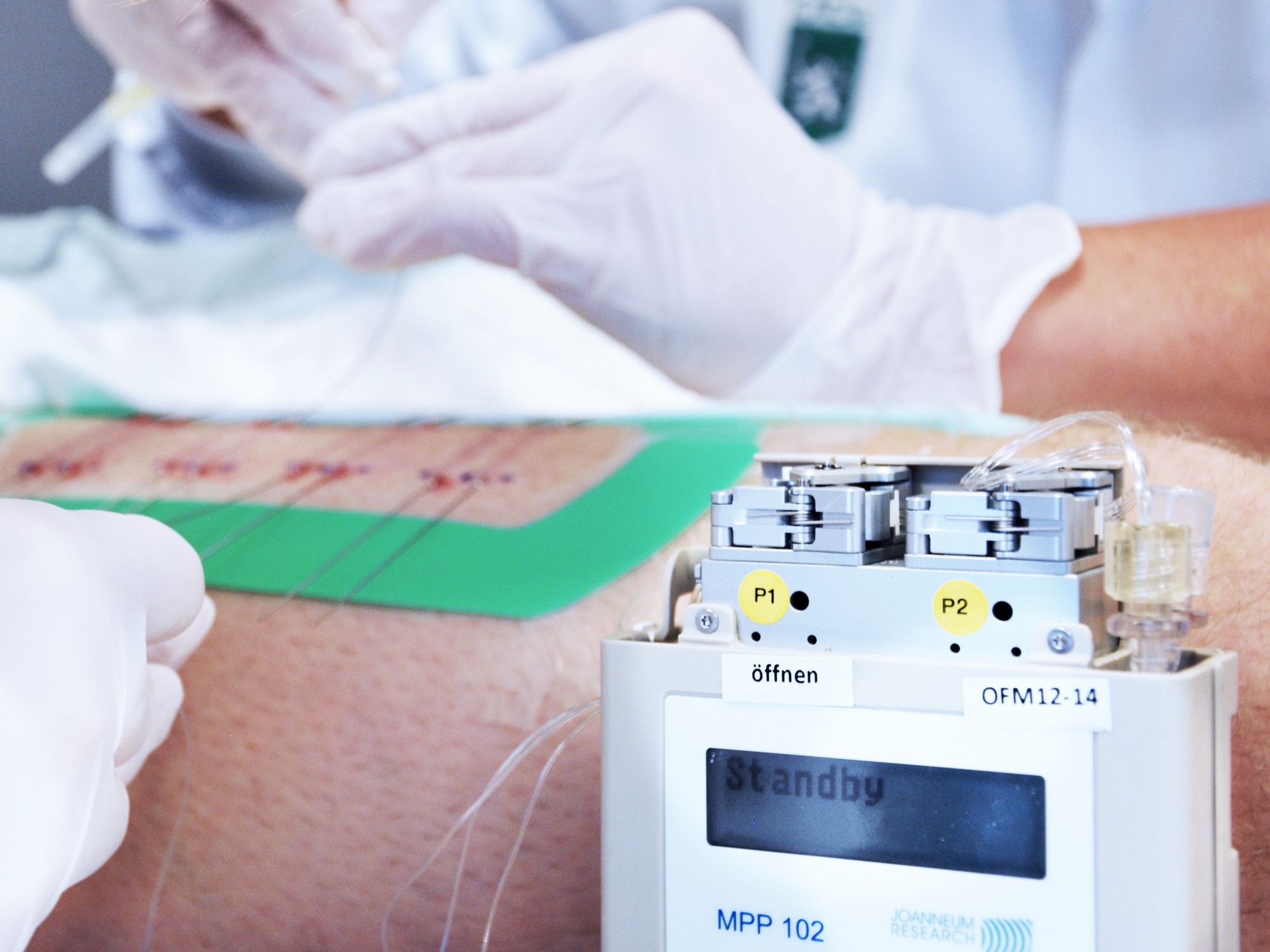
Our innovative OFM (open flow microperfusion) sampling technology is a valuable tool for PK-PD studies in neuroscience, dermatology, oncology, and biomarker research. Minimally invasive, membrane-free OFM probes enable access to the entire biochemical information of the interstitial fluid from brain and peripheral tissues, and thus opens up new horizons in preclinical and clinical drug testing.
 Efficient
Efficient
OFM studies reduce costs by providing a complete pharmacological profile early in drug development.
 Targeted
Targeted
OFM probes enable continuous sampling from clearly specified tissue types.
 Universal
Universal
OFM samples all substances from the interstitial fluid independent of size, lipophilicity or protein-binding.