cOFM Frequently Asked Questions
1. cOFM Probe
1.1. What difference between a microdialysis guide cannula and the cOFM guide do I need to consider when calculating the ventral stereotaxic coordinates for my region of interest?
Ventral stereotaxic coordinates have to be recalculated with respect to the open length of the cOFM guide and the spatial distribution (size and form) of the region of interest. The shaft length has to be chosen in a way that the open length is inside or immediately adjacent to the region of interest after implantation of cOFM guide.
1.2 How do I order a custom probe cOFM-P-X-Y?
Contact Joanneum Research for custom-made probes. Please note that customized probes may differ in price and delivery time. Many brain regions can be addressed by standard probes. However, if a custom probe is needed, they should follow the cOFM scheme:
cOFM-P-X-Y:
Y should always be 1 mm or 2 mm.
X should be between 1 mm and 20 mm +/- 0.2 mm
Probe performance is guaranteed only for standard probes.

1.3. When I change the healing dummy against the sampling insert, is there a run-in period like with microdialysis where the membrane has to swell?
Unlike microdialysis, OFM has no membrane and therefore, no membrane swelling step is necessary. However, we suggest to perform a run-in phase at the sampling flow rate for at least one hour before starting sampling to achieve a stable flow rate (refer to the Instructions of Use for the cOFM probe).
Take care that the sampling insert and tubing are free of air bubbles before inserting them into the guide.
1.4. Can cOFM probes be autoclaved?
No, cOFM probes are gamma irradiated before delivery and autoclavation is not recommended. cOFM probes might be destroyed during autoclavation due to high temperatures.
1.5. Is there a cap that goes over the healing dummy?
No, there is no cap for the healing dummy.
1.6. These pieces are so small! How is it possible to do this in an awake animal?
For implantation, animals have to be anesthetized with appropriate anesthetics (e.g. isoflurane or ketamine) for 30 to 45 min. Then let the BBB recover for 14 days.
To replace the Healing Dummy with the Sampling Insert animals have to be sedated again. After removing the Lock, the Healing Dummy can be carefully removed from the OFM guide and replaced by pre-flushed Sampling Insert. Afterwards secure it with the Lock. Taking account the size of the pieces, Joanneum Research recommends the use of tweezers. Also, refer to the user manual of the cOFM probe, that can be downloaded from www.openflowmicroperfusion.com/info.
1.7. Do I have to remove and clean the healing dummy during the healing process?
No, cleaning during the healing process is not necessary. We recommend keeping the healing dummy in place during the whole healing process until it is exchange with the sampling insert. Removing the healing dummy during the healing process may cause additional or new injuries and thus prolong the healing process.
1.8. I want to use OFM probes with MRI. Is there metal anywhere in the probes?
No, cOFM probes are manufactured of PEEK and are not containing any metal, and can be scanned using MRI. But take into consideration that probes are attached to the scull using metal screws.
1.9. I need a step-by-step instruction on how to implant and handle the cOFM probe.
For details, refer to the user manual of the cOFM probe, that can be downloaded from www.openflowmicroperfusion.com/info.
2. OFM-Pumps
2.1. Does your pump integrate push and pull options in the same unit? Is there any calibration required?
Yes, the OFM pump (MPP102PC) has push and pull options in the same unit. Either one pump head can be used for push and the second pump head for pull or, with special tubing (e.g. OFM-PP1-LT-1000), push and pull modes can be performed using only one pump head.
Calibration is necessary as different tubing materials and diameters are available. The calibration factors are pre-determined to save you time and money and improve your research quality. The calibration factor are stated on the label of the packaging. You can find more details in the MPP102PC user manual.
2.2. How tightly controlled are the push and pull pump heads? Is there any difference between the push and pull pump head settings (for example, pull setting is 10% higher than the push)?
Flow rates can be adjusted between 0.1 – 10 µl/min with an accuracy of less than ± 20%. We recommend using pre-calibrated tubing to ensure sufficient accuracy. Furthermore, we suggest having the same flow rate for PUSH and PULL to avoid altering the pressure within the target tissue of the experimental animal.
2.3. What is the flow rate range with which I can operate the OFM pump in push-pull mode?
Lowest flow rate: 0.1 µl/min
Highest flow rate: 10 µl/min.
Flow rates can be adjusted between 0.1 – 10 µl/min with an accuracy of less than ± 20%.
We recommend using pre-calibrated tubing to ensure sufficient accuracy.
2.4. Can I install the three channel tubing to the pump and not use two of the channels?
Yes, we recommend using the central channel to ensure high sampling quality if you are not using all three channels. If a perfusate bag is used, unused channels need to be pinched off directly after the perfusate bag luer secured connector by pinch clamps. To reduce user errors, we recommend the use of single channel tubing to operate one single probe and the use of three channel tubing to operate up to three probes.
2.5. Is the battery recharging when the AC is plugged in?
No, the battery does not recharge. But if the pump is operated on power supply the battery will not be discharged because it serves as a buffer battery in case the power supply fails. If the pump is not used for more than one week, remove the battery to avoid damaging the pump by battery leakage.No, the battery does not recharge. But if the pump is operated on power supply the battery will not be discharged because it serves as a buffer battery in case the power supply fails. If the pump is not used for more than one week, remove the battery to avoid damaging the pump by battery leakage.
2.6. Is there any potential for CPU control of the pumps in the future?
JOANNEUM RESEARCH is considering a CPU control for the next version but it is not yet available for the current version.
2.7. How do you fit up to three tubings at one pump head?
There are tubing available from JOANNEUM RESEARCH that support operating up to 3 probes in parallel (e.g. OFM-PS3-75, Three Channel Push Tubing or OFM-PL3-75 Three Channel Pull Tubing). Please refer to the OFM pump user manual, which is available on the JOANNEUM RESEARCH webpage.
2.8. If we can use three tubings at one pump head, is it possible to use them on three separate animals?
In theory, using three tubings at one pump head allows you to use three separate animals, as long as you are able to achieve a stable sampling process. However, an excessive tubing length might be counterproductive to achieve stable sampling. Therefore, Joanneum Research suggests one pump for three channels only for ex vivo experiments, whereas for animal studies we suggest using one pump for each animal.
2.9. What is second flush mode? Is this necessary when running on one flow rate?
Flush mode is generally used to flush the tubing and get rid of air bubbles. Thus, the pump is operated at a freely selectable flowrate of up to 10 µl/min. The flush mode allows you to select the amount of time and the flowrate at which the system will be flushed and what happens after the time has elapsed e.g. "Auto Pump", "Confirm Pump, "Auto Stop", "Confirm Stop". Please refer to the OFM pump user manual available on www.openflowmicroperfusion.com/info.
The second flush is identical to first flush mode, but allows you to set different parameters. This might be necessary for some experiments.
In general, we recommend to flush the tubing until it is air bubble free and then to perfuse it at the desired flow rate of e.g. 0.3 to 1.0 µl/min.
2.10. Why would we need to have different flow rate settings for P1 and P2?
cOFM probes are normally used at the same flow rate for P1 and P2 (e.g. 0.3 to 1 µl/min). However, as the pump is also designed to operate dermal probes, flow rates might vary depending on the experiment. Joanneum Research recommends operating dermal probes
(a/d OFM-P-15) with a slightly higher PUSH- than PULL-flow rate e.g. 1.1/1.0 µl/min.
2.11. If we have to adjust the tubing adj %, does it automatically reflect on the flow rate?
Tubing are precalibrated, thus the flow rate shown on the pumps display reflects the true flow rate if the “Tubing Adj %” is set to the values shown on the packaging.
2.12. Should the pump work with just a battery?
Yes, pump can work with just a battery.
Typical operating time (with a new battery) is up to 48 hours at 1.0 μl/min and up to 24 hours at 10 μl/min. For longer operating time, change of battery is necessary or operation with power supply. Please refer to the OFM pump user manual available on www.openflowmicroperfusion.com/info.
3. Perfusate/ Perfusate Bag
3.1. Is there any loss of perfusate volume in the brain when using the cOFM probe with the OFM pump or some other peristaltic pump?
Volume loss is possible if the push rate is higher than the pull rate. For proper cOFM sampling, we highly recommends the use of an OFM pump, which has guaranteed flow rate accuracy. Alternatively, syringe pumps can be used, as they have a comparably accurate flow rate over time.
3.2. Do I have to use the OFM Bag to deliver the perfusate (e.g. aCSF) or can I use a container filled with the buffer externally?
For better sampling results, we recommend using the OFM perfusate bag, which is delivered fully sealed in gamma irradiated protective packaging. However, if necessary, you can use any container filled with any perfusate.
3.3. Do I need to include BSA (Bovine Serum Albumin) in the perfusate to minimize non-specific binding? What percentage do you recommend?
The perfusate composition is crucial for successful cOFM sampling. We suggest performing an in vitroadsorption test prior to the in vivo sampling to assess the degree of unspecific adsorption of the substance of interest to the cOFM system.
3.4. How do you prevent liquid coming out of the connectors on the perfusate bag when filling with a syringe?
Please refer to the user manual of the perfusate bag. It shows in a step-by-step manner how to install and fill the perfusate bag in an air bubble free way.
1. Unpack tubing and perfusate bag.
2. Remove red protection cap of perfusate bag by turning it counter clockwise.
DO NOT remove needleless injection port.
3. Connect perfusate bag with the push tubing by turning the luer-connector clockwise.
4. Fill the perfusate bag with a (sterile) syringe via the needleless injection port.
CAUTION: DO NOT use aggressive fluids (alcohols, acids, bleaches, etc.) or hot fluids
(> 45°C) as perfusate (see instructions for use of perfusate bag).
4. Tubing
4.1. What is the difference between single channel and three channel tubing?
With single channel tubing, one single probe can be operated. Three channel tubing allow operating up to 3 probes simultaneously.
4.2. Can we use the same tubing that we use for microdialysis with OFM?
No, other tubing than the ones developed for OFM will damage the OFM pump.
4.3. Does the entire tubing need to be switched after every use? In the past, I have used the same tubing for 5 continuous days (usually 3 to 4 studies that are 24 hours long with overnight stabilization and a buffer flush for a few min, after disconnecti
No, the tubing does not have to be switched after every use.
The tubing are gamma irradiated, but biofouling can still occur after using the tubing for a prolonged period of time, especially under unsterile conditions. Furthermore, tubing wear out over time, but can be typically used for 72 hours. If tubing are used longer, check the flow rate by measuring sampling time and volume and calculate the actual flow rate (flow (µl/min) = volume (µl) / time (min). If the tubing is not used for a couple of days between two sampling sessions, open the pump head and remove it from the pump. If multiple sampling sessions in experimental animals are performed, we recommend one day of rest for the experimental animal to recover. For every sampling session, use a lock to secure the sampling insert and use new tubing systems to guarantee the best sampling quality.
4.4. Are there any special requirements when cutting the tubing to length (razor/scalpel)?
We recommend using a scalpel or a brain matrix blade with a 45° angle.
4.5. Is there only one length of the low bind tubing?
No, the low bind tubing OFM-T1-200-LB and OFM-T2-100-LB come in standard length and can be shortened depending on your needs.
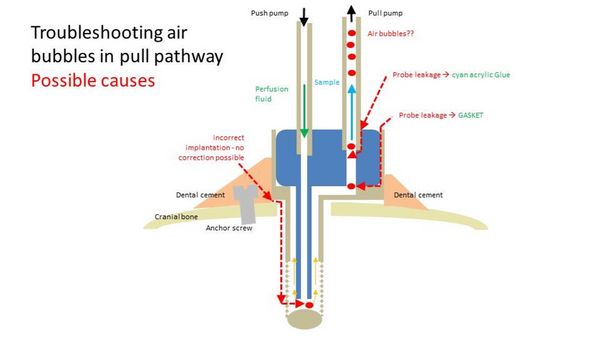
4.6. I found air bubbles in my tubing. What can I do to get rid of them?
Air bubbles can enter the tubing system due to the following three reasons:
(Also refer to the following picture)
1.) The Guide was not properly attached to the cranial bone of the animal or was loosened during manipulation (e.g. while inserting the Sampling Insert). In this case, re-implantation is necessary.
2.) The Sampling Insert leaks. In this case, check if the gasket is in place.
3.) Air bubbles enter between tubing and Sampling insert. In this case, use a drop of cyan acrylic glue.
5. Sampling Process
5.1. Does the cOFM sampling insert cause damage to brain tissue while sampling?
No, the cOFM sampling insert is not causing damage to the brain during sampling. But, the shaft of the cOFM guide causes a trauma and a disruption of the blood-brain barrier during implantation. After implantation, the healing dummy inside the cOFM guide prevents ingrowth of brain tissue during blood-brain barrier re-establishment and trauma abatement. For sampling the healing dummy is replaced by the sampling insert. Unlike in microdialysis, where the membrane is longer than the guide (stylet) and might cause additional damage during insertion, the cOFM sampling insert is shorter than the guide and thus causes no addional damage (it is moved inside a cage).
5.2. Can I sample neurotransmitters with cOFM?
Yes, cOFM allows sampling of hydrophilic to lipophilic substance regardless of molecular size. However, the whole setup (open length, flow rate, perfusion fluid, and additional materials in the system like connectors) has to be adapted to achieve optimal results.
5.3. Is there a recovery factor for cOFM probes? What are the factors that influence recovery of molecules through cOFM probes in vivo? Is dilution of ISF a factor?
cOFM can be calibrated (= recovery estimation) for absolute quantification of substances in the brain. Influencing factors are the size of open length, flow rate, and perfusate composition. Additionally, the physical characteristics of the substance of interest can influence the recovery factor (e.g. adsorption to components of the cOFM system). ISF that is sampled by cOFM is diluted, but the overall ISF composition is left unchanged.
5.4. What is the principle of in vitro recovery in cOFM?
The in vitro recovery of cOFM assesses the adsorption of the substance of interest to the cOFM system. In vitro recovery tests should be done with the same system components that are used in the in vivo setup (cOFM probe type, tubing, etc.). Once the adsorption is assessed, in vitro recovery can be used as a correction factor for in vivo results. In vitro recovery cannot be used as an in vivo recovery surrogate.
5.5. What is the best way to calculate in vivo recovery using cOFM probes?
In vivo recovery is empirically assessed. Commonly used protocols can be found in the literature (e.g. no net flux method, retrodialysis etc.). The suited in vivo cOFM probe calibration method should be chosen according to the needs of the experiment.
5.6. We are interested in sampling in awake animals at different time points over several days (maybe two samples per day), but we do not need to use the perfusate for the entire period. Is it necessary for the pump to be continuously operated between the
The cOFM sampling insert can be replaced with the healing dummy after sampling, if you are not continuously perfusing the cOFM system. Leaving a sampling insert in the guide without continuous pumping may lead to clogging. cOFM sampling can be restarted on the next sampling day by following the protocol in the user manual. This procedure can be repeated as long as the cOFM probe and animal are in good condition.
We recommend one day of rest for the experimental animal to recover. For every sampling session, use new tubing systems to guarantee best sampling quality.
5.7. I understand that the pump can be used for up to one month of continuous sampling, but how long can I continuously perfuse in vivo? Is there any evidence of tissue reactivity caused by the pump liquid flow into the cOFM probe/brain?
Typical continuous sampling sessions last from 24 h to 5 days. There is no evidence of tissue reactivity. We suggest changing the tubing system after 72 hours. If tubing are used longer, check the flow rate by measuring sampling time and volume and calculate the actual flow rate (flow (µl/min) = volume (µl) / time (min). For more information, please contact Joanneum Research
5.8. When hooking up animals should the sampling unit be flowed overnight? If not, how much flow in the morning is typical before collections should start?
For OFM sampling, overnight flow rate stabilization is not required. You can insert the sampling insert in the morning. Afterwards we recommend to flush the tubing until it is free of air bubbles and afterwards to perfuse it with the desired flow rate of e.g. 0.3 to 1.0 µl/min.
5.9. When performing multiple sampling experiments over time with the same animal, is it necessary to replace with the healing dummy between experiments?
We recommend replacing the healing dummy when it can be visually determined to be worn, but not necessarily between two sampling sessions. We also recommend using a new sampling insert for every sampling session.
After sampling the cOFM sampling insert is replaced by the healing dummy and animals can be returned to their home cages. Before insertion, the healing dummy should be cleaned with alcohol, followed by distilled water. When working with an anaesthetized animal setup, a minimum of one day of rest is recommended before the next sampling session. cOFM sampling can be restarted by following the protocol in the user manual. This procedure can be repeated as long as the cOFM probe and animal are in good condition.
The pump should be unplugged and stopped. We suggest using new tubing for each sampling session. The tubing are gamma irradiated, but still biofouling can occur after using the tubing for a certain period, especially under unsterile conditions. Furthermore, tubing wear out over time, but they can typically be used for 72 hours. If tubing are used for a longer time period, check the flow rate by measuring sampling time and volume and calculate the actual flow rate (flow (µl/min) = volume (µl) / time (min).
6. Animals
6.1. Can I use the same animals for several intermittent sampling sessions over several days or weeks without creating tissue damage? For how long?
The healing dummy can replace the cOFM sampling insert after sampling and animals can be returned to their home cages. When working with an anaesthetized animal setup, a minimum of 1 day of rest is suggested before the next sampling session. cOFM sampling can be restarted by following the protocol in the user manual. This procedure can be repeated as long as the animal and the cOFM probe are in good condition.
6.2. Can I use the cOFM probes for rats and mice?
Yes, cOFM probes can be used for rats and mice. Standard probes are available with different lengths and customized probes can be ordered at Joanneum Research. For more information please refer to the OFM page: www.openflowmicroperfusion.com or contact us by Email: ofm@joanneum.at
6.3. Is it possible to put more than one cOFM probe in the same mouse brain?
The number of probes per animal depends on the brain region of interest and the size of the animal. When using more than one probe per animal, the surgeon has to consider the orientation of the lock, as it has to be removed when the healing dummy is replaced with the cOFM sampling insert.
7. Services/ Support/ Training
7.1. We outsource/prefer to buy animals implanted with cOFM guide from an external service provider. Will you be able to ship the guide dummy unit and the sampling insert to two different locations?
Yes, Guide dummy kit and sampling insert are available as separate parts. For more details refer to the OFM page: www.openflowmicroperfusion.com/info or contact us by Email: ofm@joanneum.at
7.2. Do you offer cOFM contract research services?
Yes, JOANNEUM RESEARCH (JR) - HEALTH offers cOFM contract research services; based on broad experience, JR offers in vivo pre-clinical studies in animals (mouse, rat and pig) for brain, adipose and skin tissue. We perform metabolic tests to investigate the pharmacokinetics of active pharmaceutical ingredients when introduced enterally, parenterally or topically. We offer ex vivo studies in explanted human skin donated by individuals undergoing plastic surgery. We investigate the adsorption and liberation of topically active pharmaceutical ingredients. We function as One-Stop-Shop that offers:
- Study design and protocol
- Preparation and submission of ethics proposals,
- Conducting pre-clinical studies (ex vivo, in vivo)
- Laboratory analysis of samples
- Data analysis and
- Reporting
JR - HEALTH is EN ISO 9001:2008, EN ISO 13485:2012 and GLP-Good Laboratory Practice certified.
7.3. Do you provide video conferencing and technical support?
Yes, video conferencing and technical support will be provided upon request. Contact us by Email: ofm@joanneum.at
7.4. How difficult is cOFM for a researcher experienced in small or large molecule microdialysis? Do I need any special hands-on training?
Special hands-on training with OFM specialists from JOANNEUM RESEARCH is recommended to improve research quality but is not a prerequisite. All products come with a self-explaining user manual.
Following training programs are offered:
- General usage and handling of cOFM equipment
- Preclinical sampling setup in pigs and rats
- Implantation training in mice and rats
- Study design development and consulting
In addition, video conferencing and technical support or individual training sessions can be arranged.
7.5. Can I try a demo pump before making a purchase?
Yes, demo pumps are available upon request please contact us by Email: ofm@joanneum.at
7.6. Do you have user manuals for OFM pumps and cOFM probes?
User manuals are available for download from the JR webpage by scanning the QR-code or entering the link into a web browser, printed on the package of the OFM products.
8. Others
8.1. Does cOFM damage the Blood Brain Barrier (BBB)?
Yes, during the implantation of the cOFM guide, the BBB is manually disrupted. Therefore, a trauma abatement and BBB re-establishment time of 14 days is recommended prior to sampling (Birngruber et al., 2013. Clin. Exp. Pharmacol. Physiol. 40, 864–71). After these 14 days, the BBB integrity of animals with implanted cOFM guide is fully reestablished and comparable to control animals without cOFM.
8.2. What will be the infusion factor?
cOFM probes can also be used as a tool for infusion of substances. For simple infusion (push only), the infusion factor is 100%. If substance or molecules are perfused (push-pull), the factor is different and depends on flow rate, open length and substance characteristics (diffusion in tissue, clearance from tissue, etc.).
8.3. When was the cOFM technology developed and how long has it been in use?
JOANNEUM RESEARCH began the development of OFM technology about 15 years ago. Due to the great success and the new demands for OFM products in clinical/ preclinical studies, JOANNEUM RESEARCH developed cOFM probes within the last few years for cerebral and neurological brain studies.

